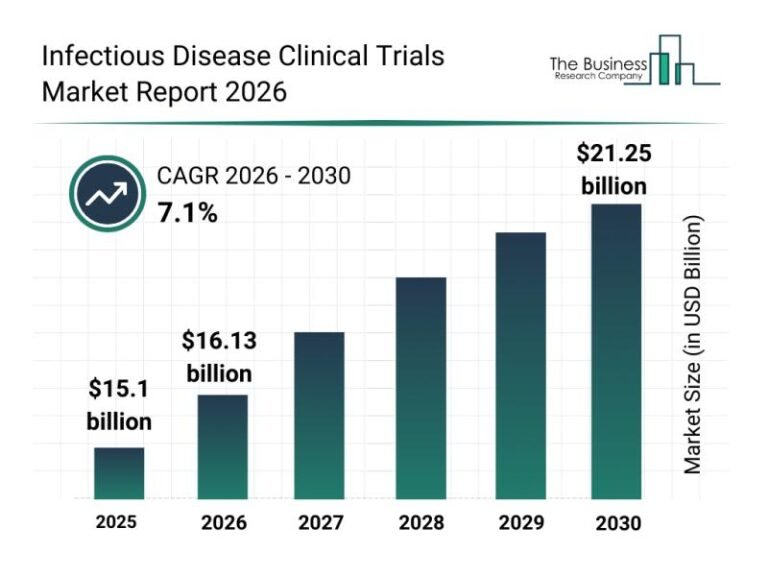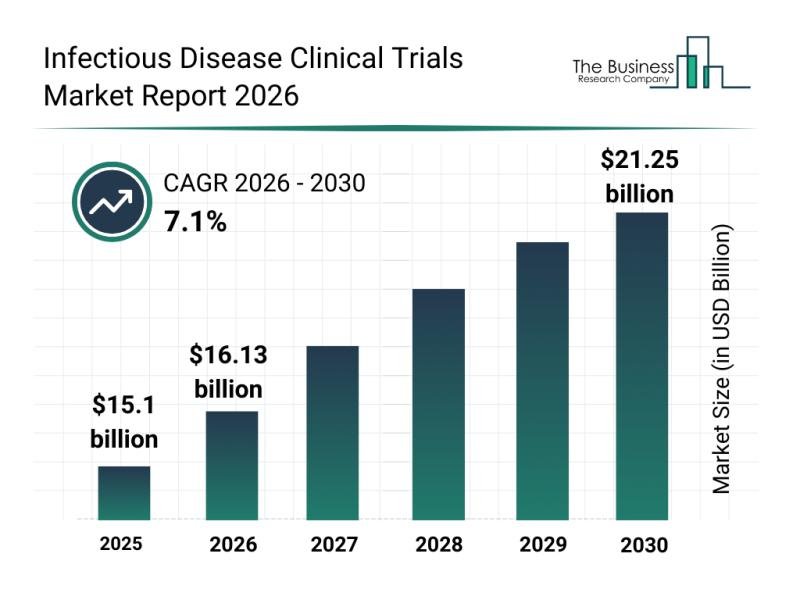
The infectious disease clinical trials growth is reshaping the medical research landscape, with significant implications for global health advances. This shift towards expedited and more efficient drug development is driving substantial market expansion. As infectious diseases present ongoing threats worldwide, the urgency to innovate within clinical trials has never been more pronounced. Therefore, understanding the trends fortifying this sector is crucial for stakeholders aiming to navigate this evolving field.
Driving Factors Behind Infectious Disease Clinical Trials Growth
Technological advancements play a pivotal role in the surge of infectious disease clinical trials. Innovations, such as digital health technologies and artificial intelligence, streamline clinical trial processes, enhance data accuracy, and reduce the time needed to bring new treatments to market. Consequently, these improvements position stakeholders to respond more swiftly to emerging infectious threats.
Investment Fuels Innovative Trial Approaches
Increased investments in research and development directly contribute to the growth of infectious disease clinical trials. With heightened financial support, researchers can pursue cutting-edge methodologies and embrace novel approaches, such as adaptive trial designs and patient-centered studies. Additionally, collaborations between pharmaceutical companies, academic institutions, and government entities amplify these efforts by pooling expertise and resources. This collaborative spirit is akin to initiatives in other healthcare domains, such as transformative surgery camps, highlighting the impact of global partnerships.
Impact of Global Health Priorities on Clinical Trials
Global health priorities are shifting towards rapidly addressing infectious diseases, thereby influencing clinical trial dynamics. For example, the COVID-19 pandemic underscored the need for agile and flexible trial frameworks. As a result, the industry has adapted by implementing decentralized trials, which minimize participant burden and access barriers while expanding research reach. These approaches exemplify how current health urgencies drive the reshaping of traditional processes. Efforts similar to these addressing other health challenges also highlight adaptable strategies in healthcare.
Competitive Strategies in a Dynamic Marketplace
Increased competition within the infectious disease clinical trials sector spurs innovation and accelerates timelines. Industry leaders seek to differentiate themselves by integrating advanced technologies and leveraging big data analytics to refine trial efficiency and efficacy. Moreover, competitive pressures compel companies to prioritize patient engagement and experience, fostering greater participation and retention in trials.
In conclusion, the landscape of infectious disease clinical trials is undergoing transformative growth. This evolution is propelled by technological and financial advances, as well as a global pivot towards addressing pressing health challenges. As the market continues to expand, the implications for innovation and public health are substantial.
Key Takeaways
- Technological innovations enhance the efficiency and speed of clinical trials.
- Increased R&D investments enable cutting-edge, adaptive trial designs.
- Global health priorities fuel the evolution of more responsive trial structures.
Medical Disclaimer
This article provides informational content and does not offer medical advice.